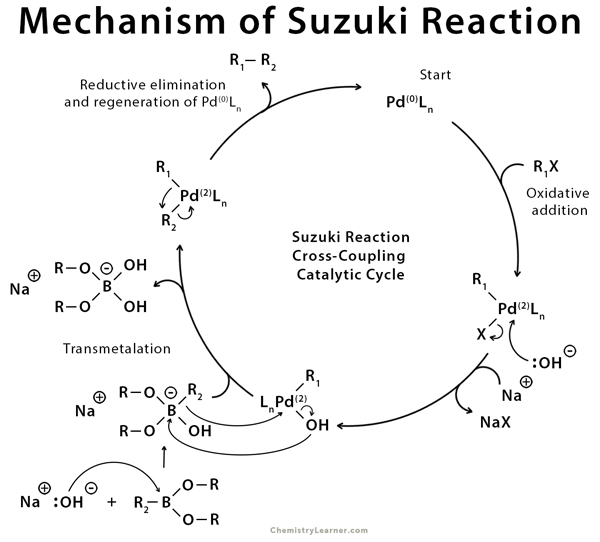
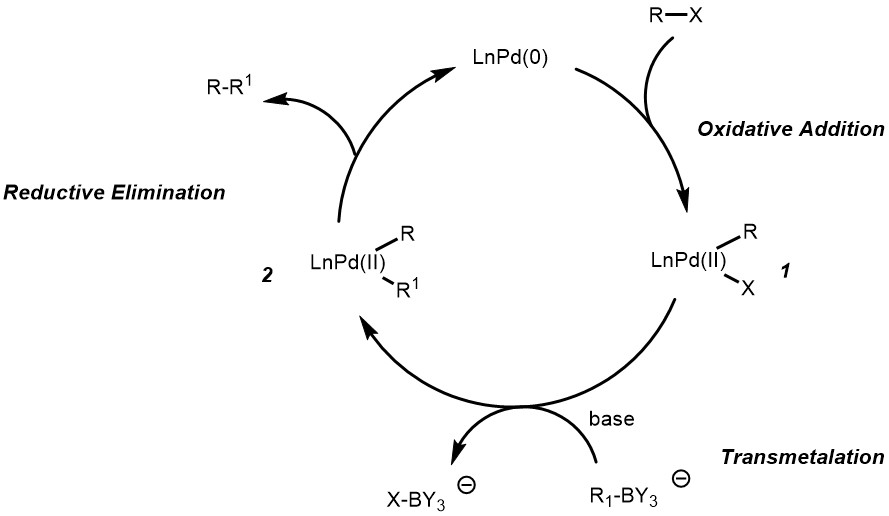
Figure 8 from Transition-metal-catalyzed Suzuki-Miyaura cross-coupling reactions: a remarkable advance from palladium to nickel catalysts. | Semantic Scholar
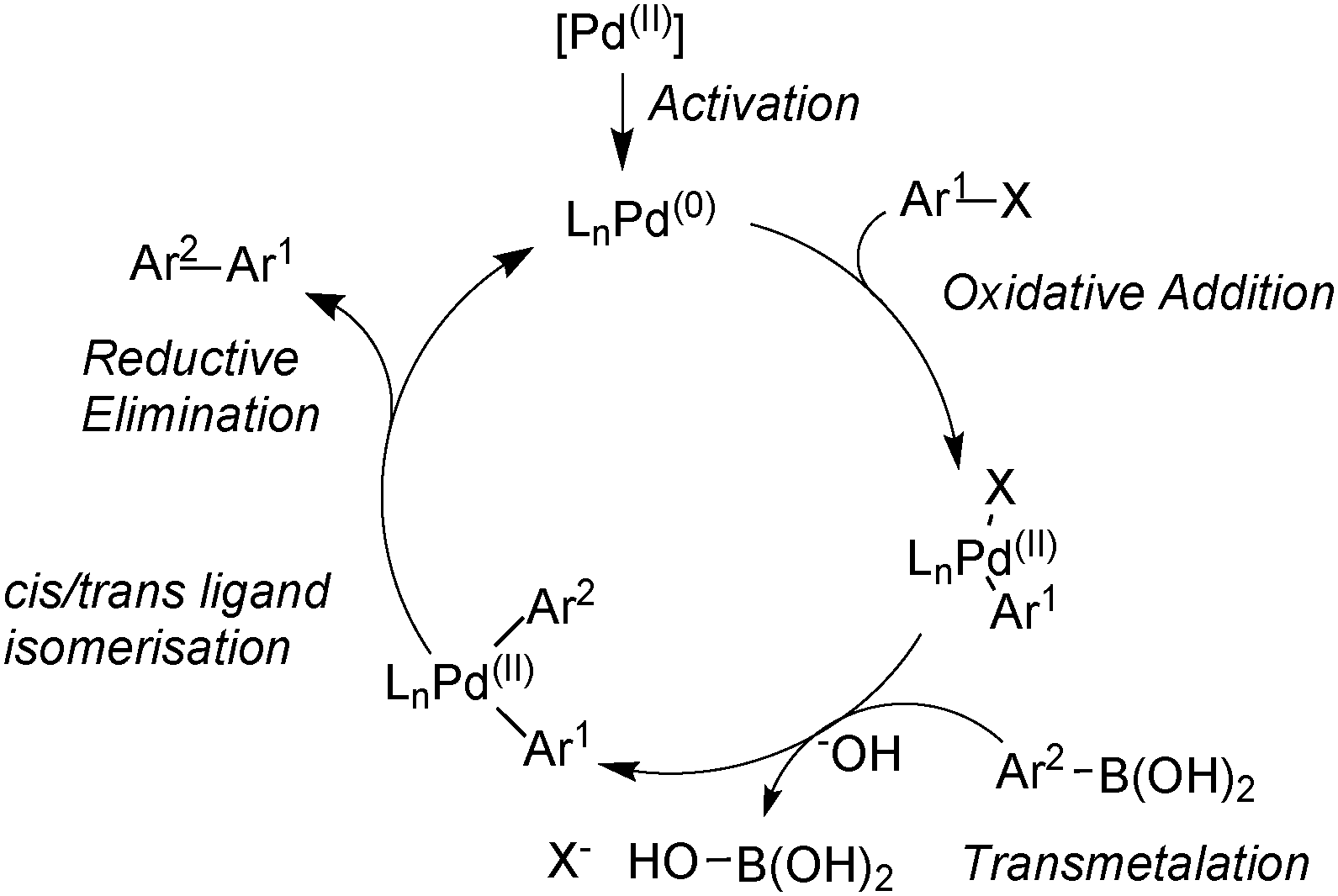
Selection of boron reagents for Suzuki–Miyaura coupling - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C3CS60197H

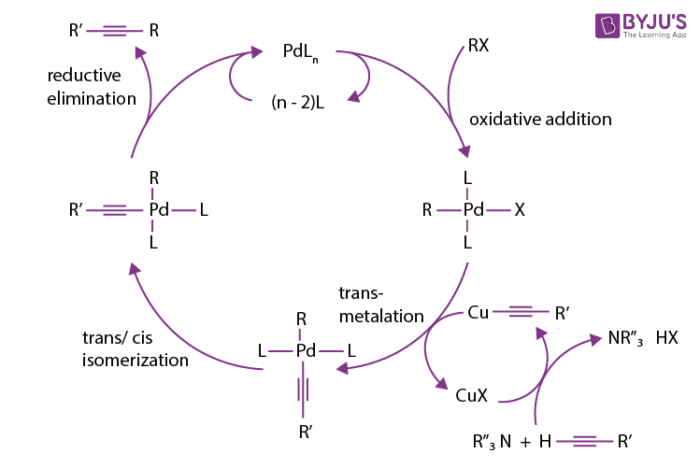













![PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar PDF] Mechanisms of Nickel-Catalyzed Cross-Coupling Reactions | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3297cc29ee21e706a719bd5da81dca59c2ac6ab2/7-Figure4-1.png)