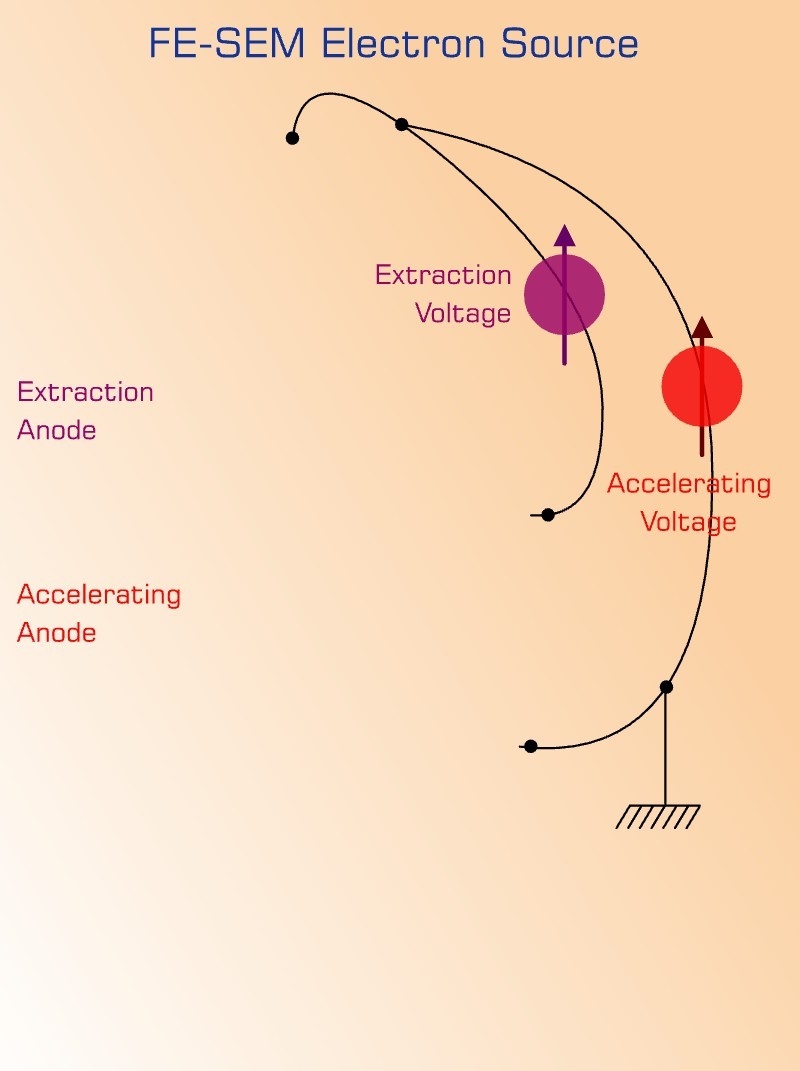
98. A: if an electron and proton possessing same kinetic energy enter an electric field in a perpendicular direction, the path of the electron is more curved than that of the proton
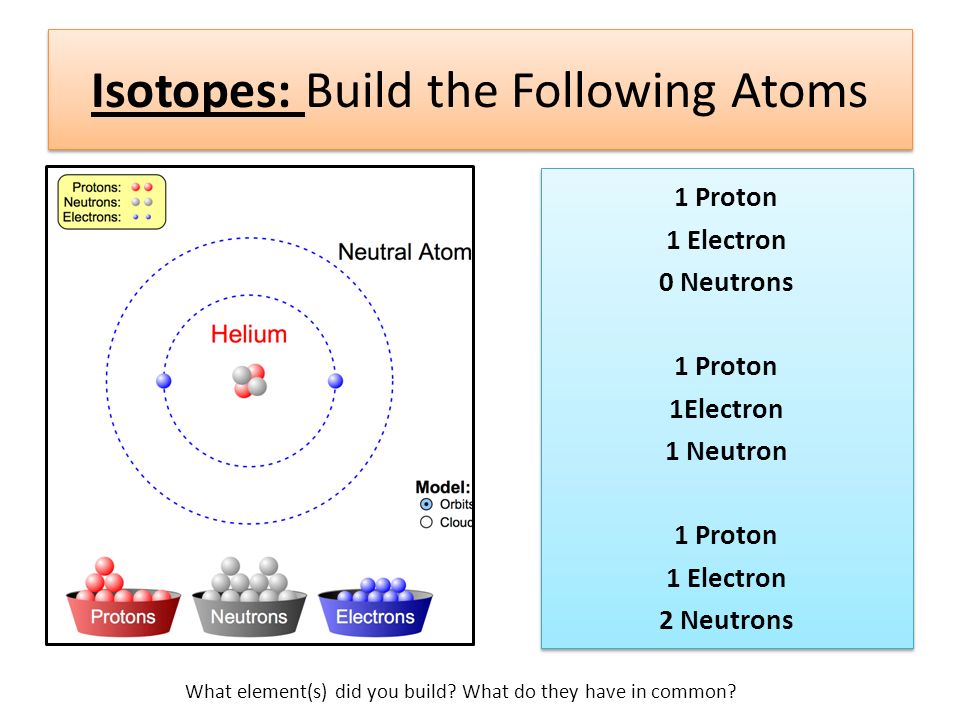
Isotopes: Build the Following Atoms 1 Proton 1 Electron 0 Neutrons 1 Proton 1Electron 1 Neutron 1 Proton 1 Electron 2 Neutrons 1 Proton 1 Electron 0 Neutrons. - ppt download

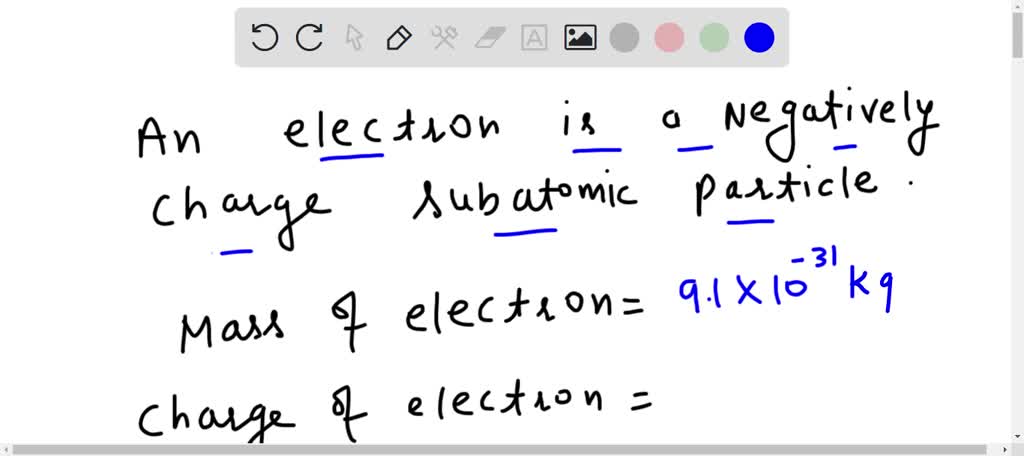
What is the radius of the path of an electron (mass 9 × 10^-31 kg and charge 1.6 × 10^-19 C ) moving at a speed of 3 × 10^7 m/s in
Structure of the Negative Hydrogen Ion Electron 1 is closer to the nucleus | Download Scientific Diagram

How to Determine the Number of Electrons on an Object Given its Net Charge in Coulombs | Physics | Study.com
energy of electron in orbit of H atom is 1.51eV. Wavelength produced by the electron in same orbit if 1st orbit of H have radius x i



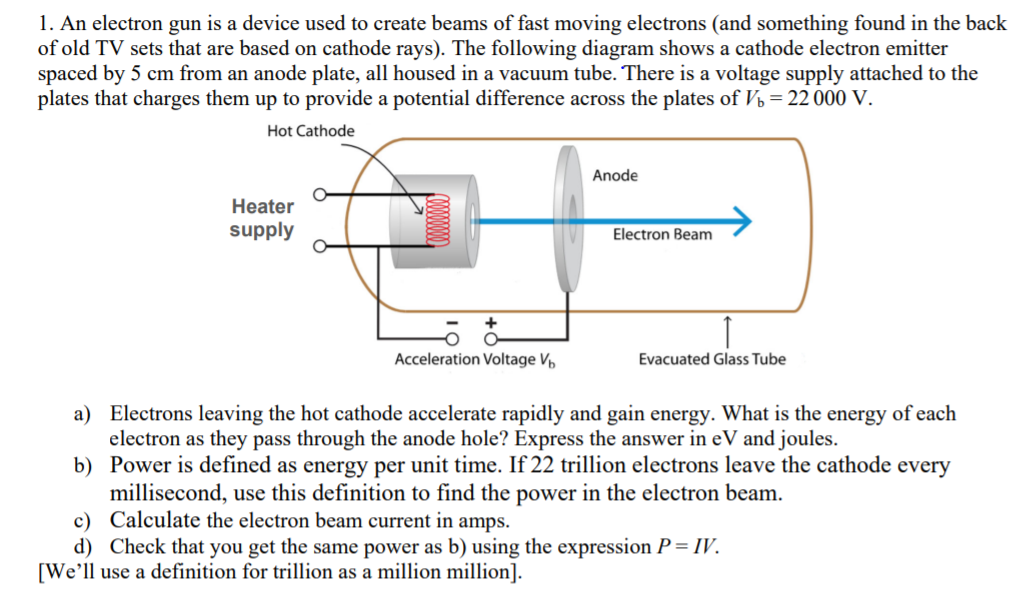
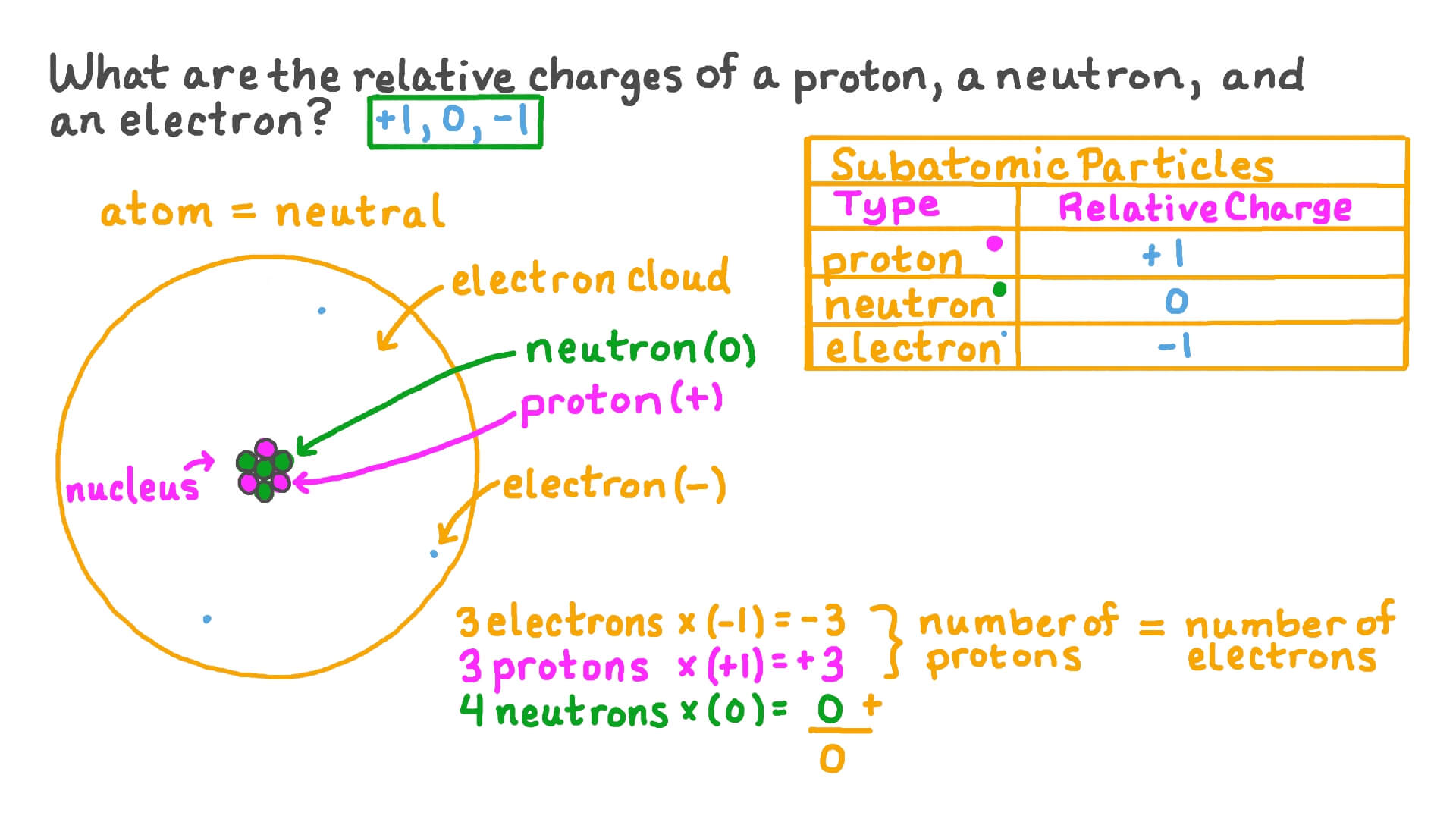


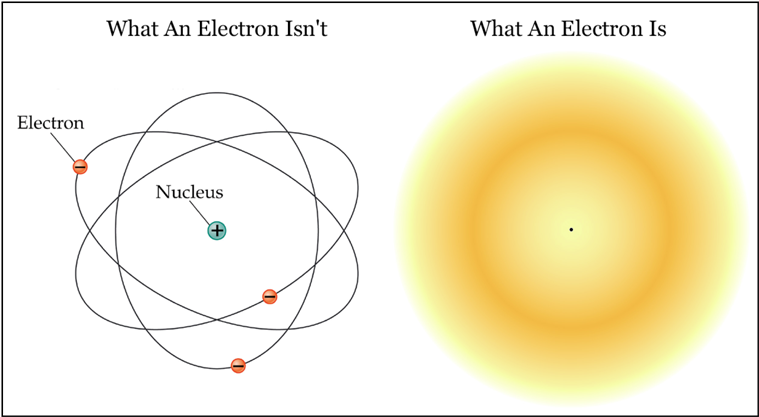
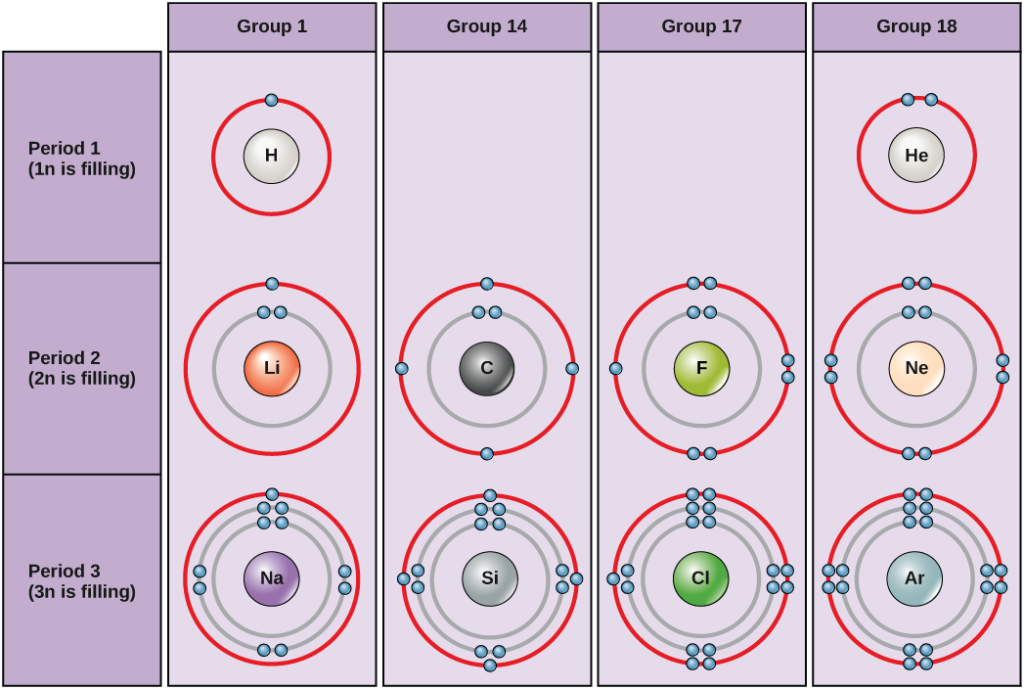
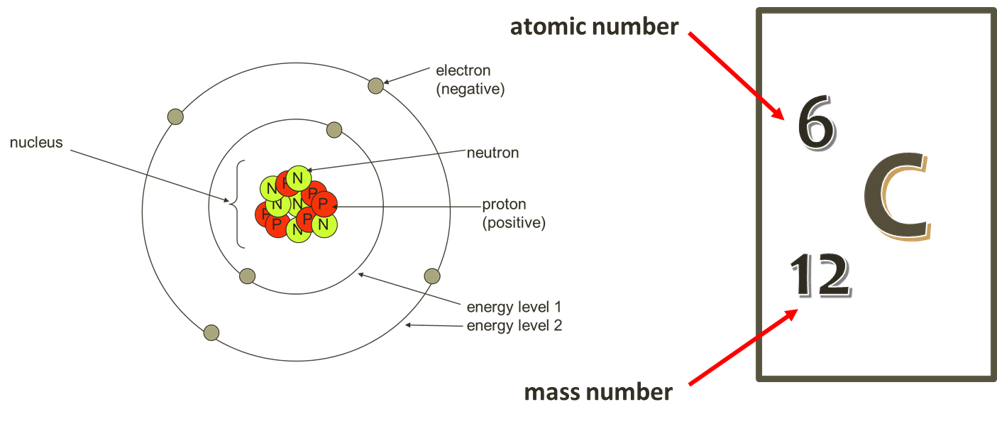
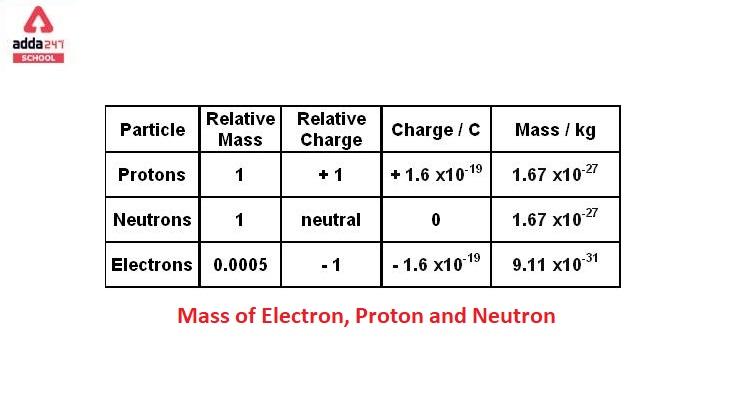


![1. Electrons in Motion - Practical Electronics: Components and Techniques [Book] 1. Electrons in Motion - Practical Electronics: Components and Techniques [Book]](https://www.oreilly.com/api/v2/epubs/9781449373221/files/assets/pect_0101.png)