Influence of the electron donor groups on the optical and electrochemical properties of borondifluoride complexes of curcuminoid derivatives: a joint ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA25436E

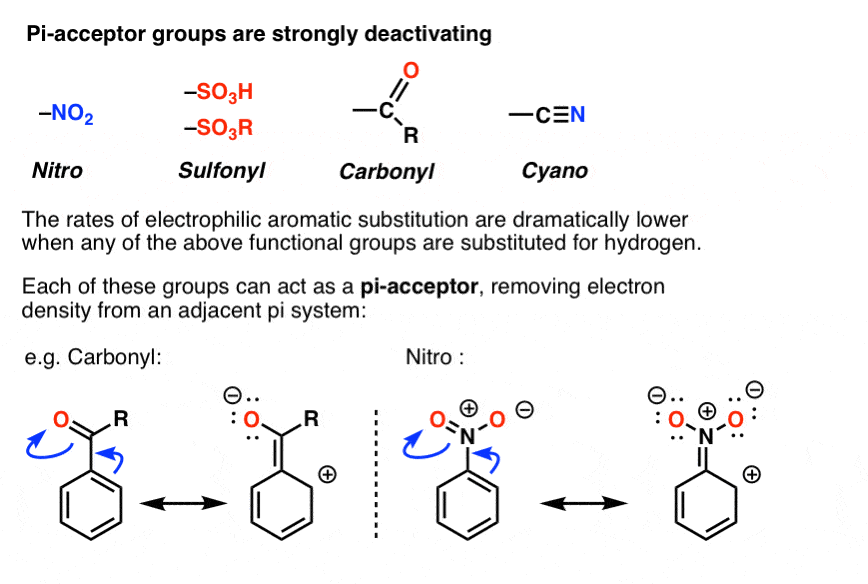
Exploring the Effect of Electron Withdrawing Groups on Optoelectronic Properties of Pyrazole Derivatives as Efficient Donor and Acceptor Materials for Photovoltaic Devices
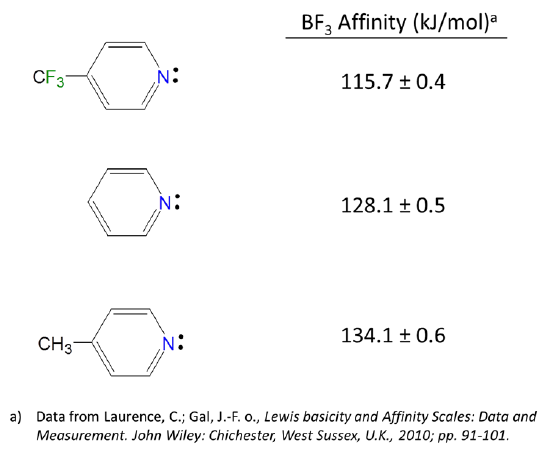
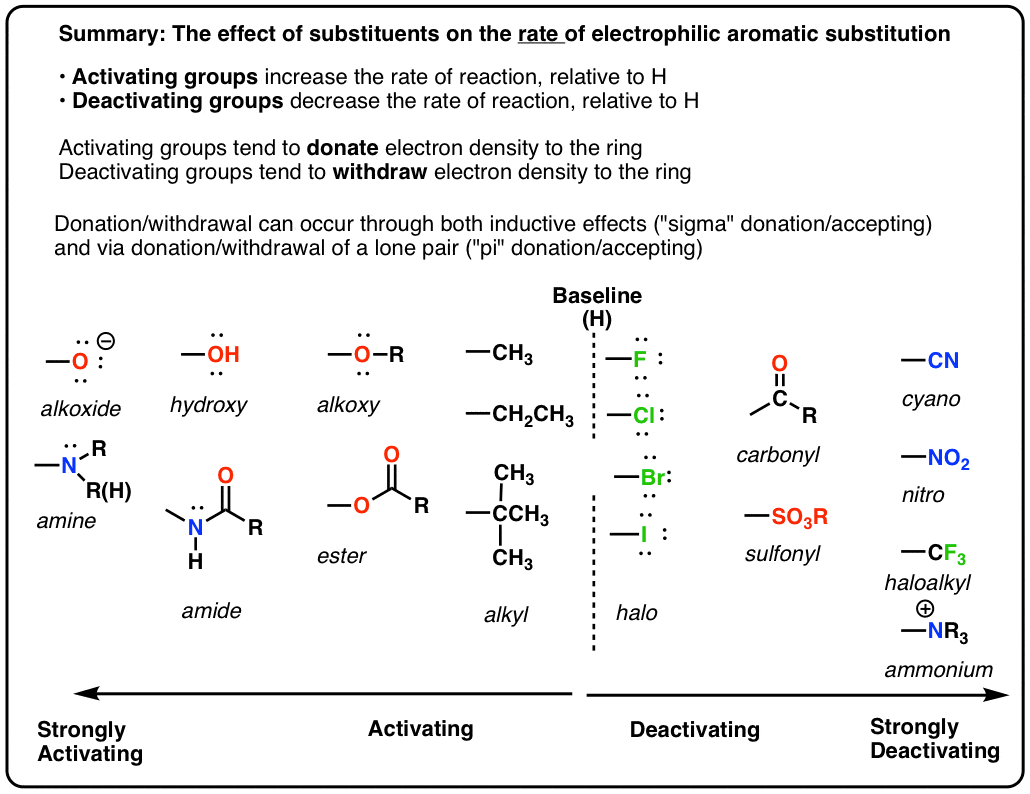
6.4.2: All other things being equal, electron withdrawing groups tend to make Lewis acids stronger and bases weaker while electron donating groups tend to make Lewis bases stronger and acids weaker -
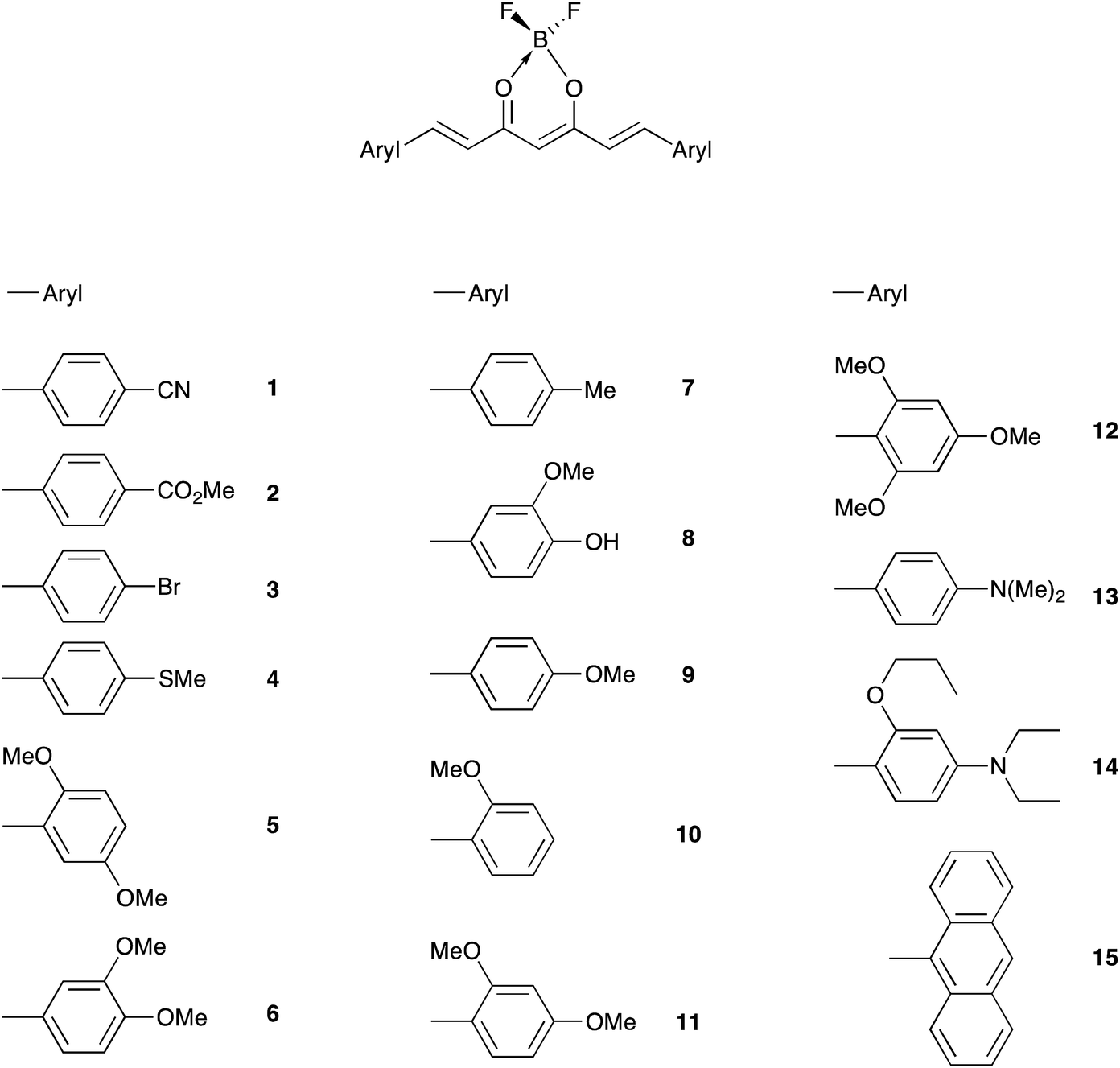
Influence of the electron donor groups on the optical and electrochemical properties of borondifluoride complexes of curcuminoid derivatives: a joint ... - RSC Advances (RSC Publishing) DOI:10.1039/C6RA25436E

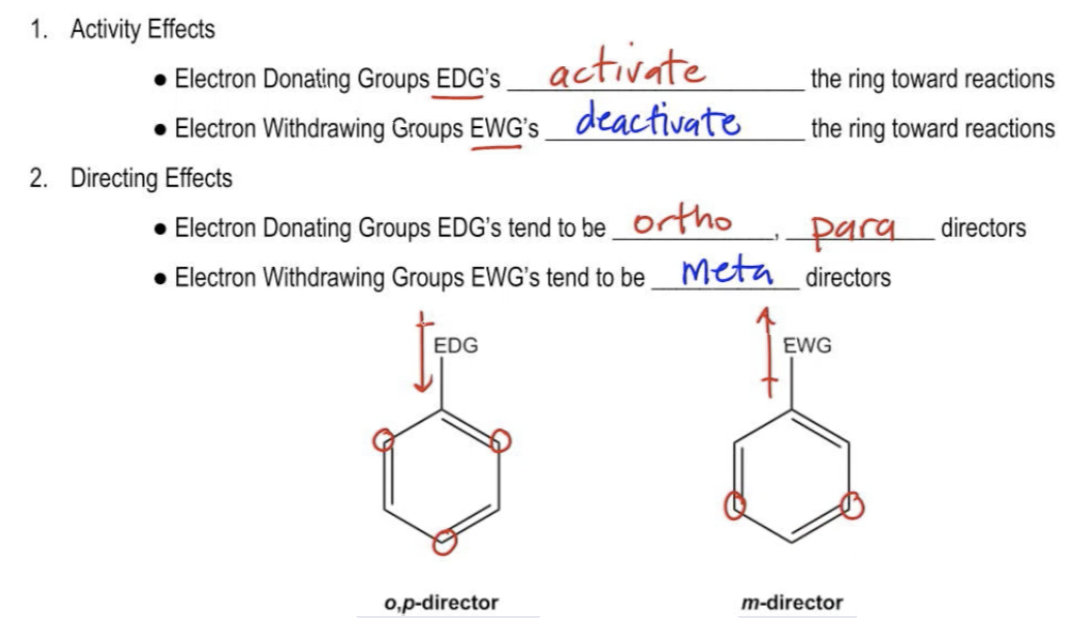
Why are ortho and para directors in aromatic rings electron donating whereas meta directors are electron withdrawing? - Quora
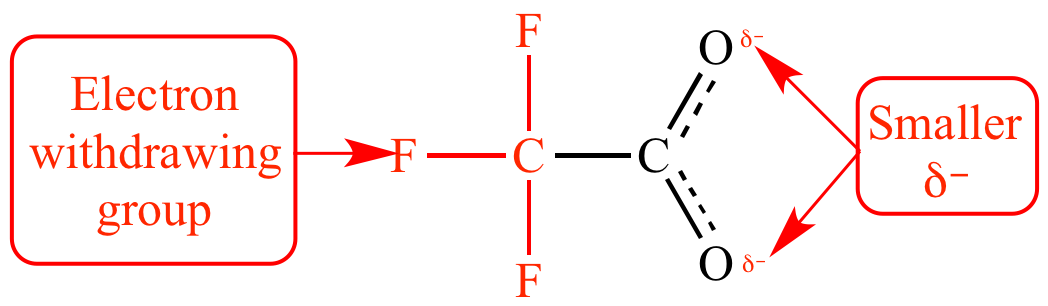
Is it true that fluorine is always the strongest electron withdrawing group (EWG) due to fluorine's unrivaled electronegativity? - Quora

organic chemistry - Amine group - electron donating or withdrawing group? - Chemistry Stack Exchange
Confusion on electron withdrawing and donating effects of alkoxy, amide, ester groups | Student Doctor Network
![Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y](https://toppr-doubts-media.s3.amazonaws.com/images/2140650/bcc67e75-4fb0-4420-aaef-47cd4d3dbd3e.jpg)
Study the following sequence of reactions and identify the product (Y) . CH3CHO + HCHO [heat]dil.NaOH X [H3O^ + ]HCN Y