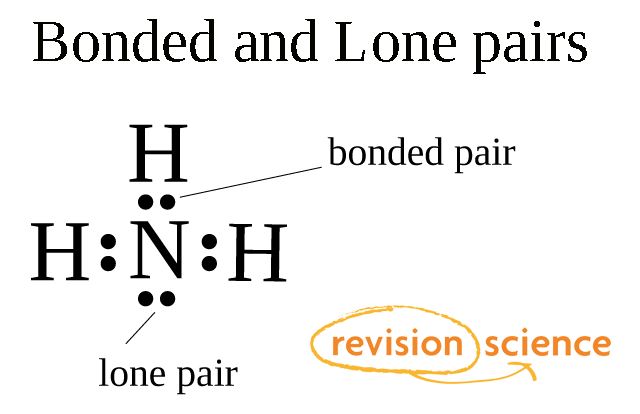Lone-pair electrons induced anomalous enhancement of thermal transport in strained planar two-dimensional materials - ScienceDirect

Identify all nonbonding lone pairs of electrons in the following molecule, and tell what geometry expect for each of the indicated atoms, the sulfur atom in the amino acid methionine. | Homework.Study.com

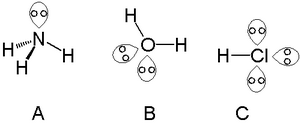
The H - C - H bond angle in CH4 is 109.5. Due to lone pair repulsion, the H - 0 - H angle in H2O will:

Find the number of molecules having two lone pair of electrons on central atom. I 3+, XeF 2, XeF 4, H 2 O , NH 2 , H 2 S , H 2 SO 4, NF 3

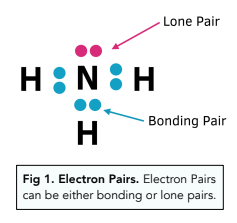
what are lone pair and shared pair of electrons experts please explain with the help of example - Chemistry - Chemical Bonding and Molecular Structure - 5672569 | Meritnation.com