Explanation origin of Colour Theory of Transition metal ions splitting of 3d level complexes electron configurations, variation of complex ion colour GCE AS A2 IB A level inorganic chemistry revision notes

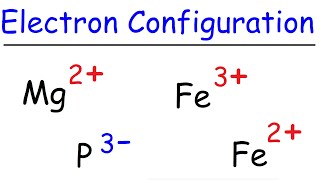
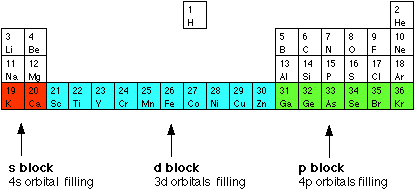
Chapter 15: Transition Metals 15.1 General Properties of Transition Metals 15.2 Complex Formation and the Shape of Complex Ions 15.3 Coloured Ions ppt download

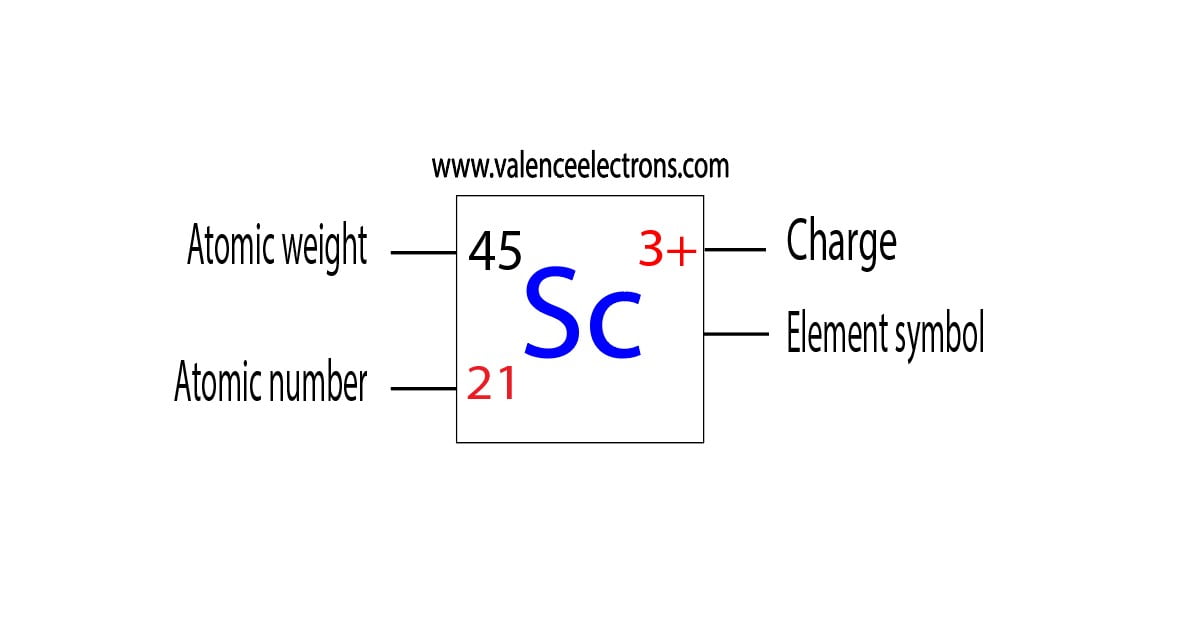
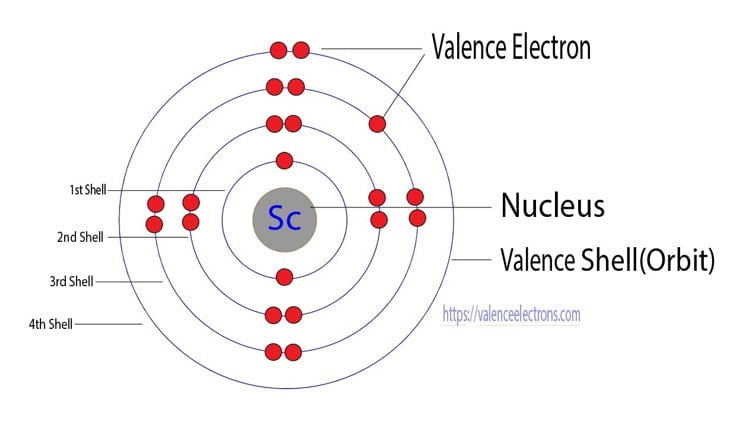
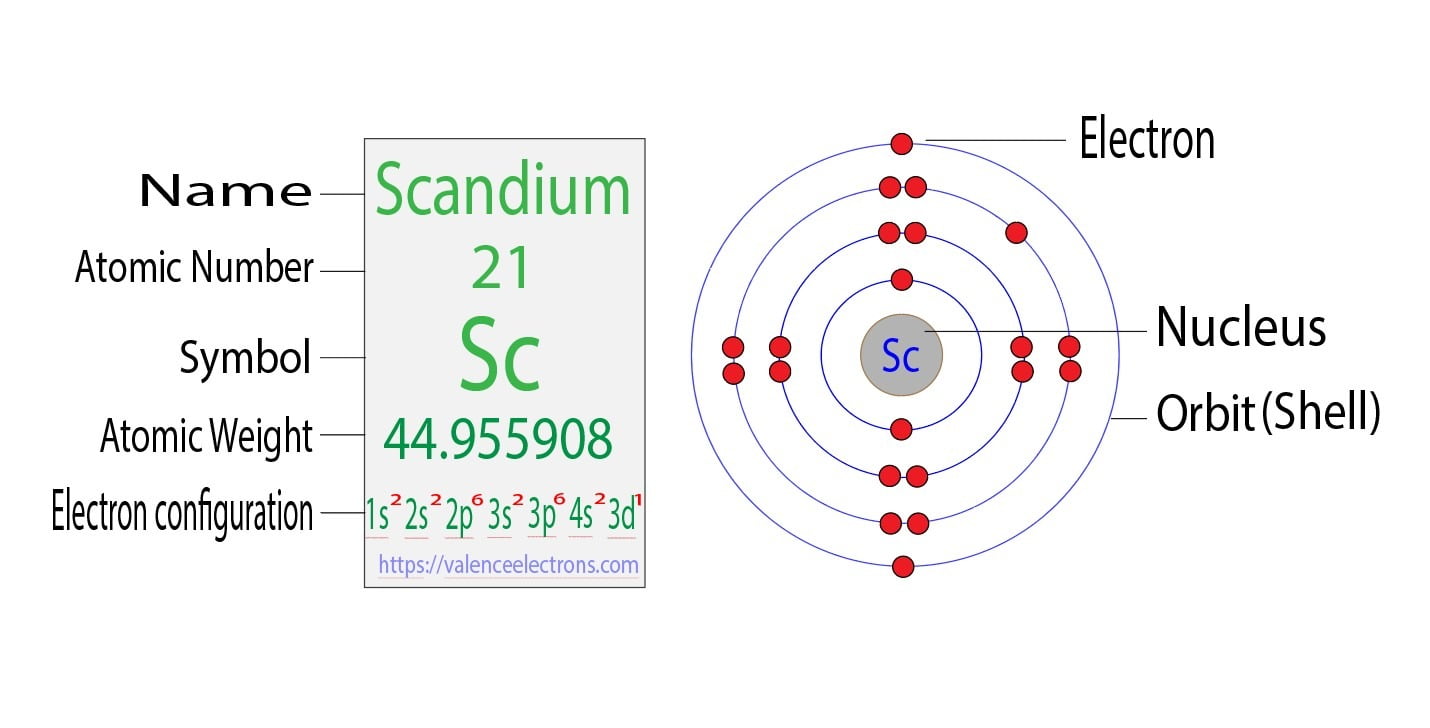
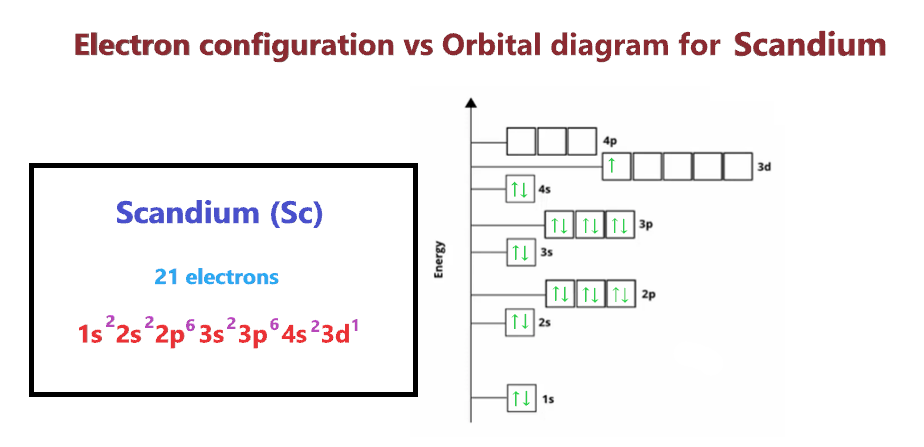
Using an orbital diagram, determine the number of unpaired electrons in scandium. | Homework.Study.com