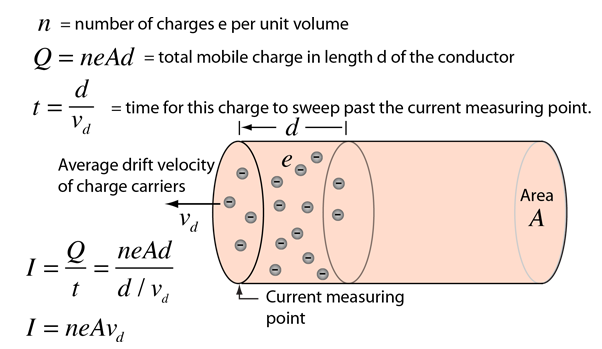
Calculate velocity of electron in first Bohr orbit of hydrogen atom (Given r = a0). - Sarthaks eConnect | Largest Online Education Community

the velocity of an electron placed in 3rd orbit of h atom will be ans is 729 107 cm s its 10 raise to 7 give me its solution ukbsx511 -Chemistry - TopperLearning.com
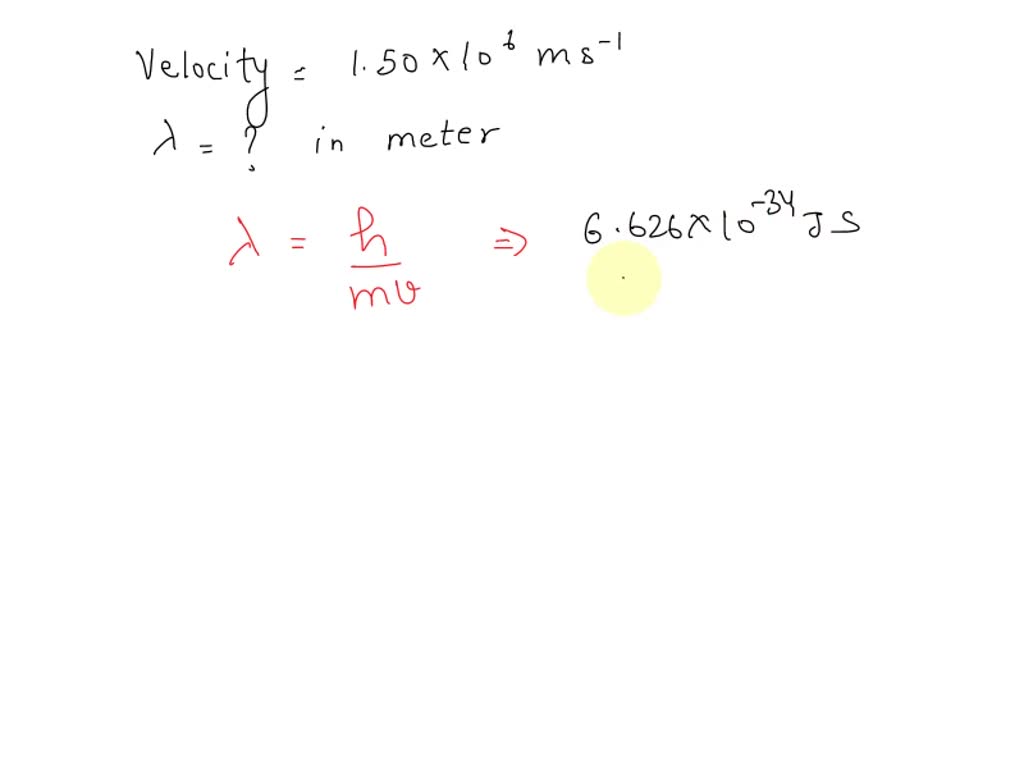
SOLVED: The velocity of the electron in the ground state of the hydrogen atom is 1.50×106 m/s. what is the wave length of the electron in meters?
Calculate maximum angular velocity of the electron in stable orbit of a hydrogen atom. - Sarthaks eConnect | Largest Online Education Community
Velocity of electron in the nth bohr orbit of hydrogen like atom is given by 2.18×10raise to power 8Z/n cm/s the velocity of electron in the first bohr orbit of hydrogen atom ? (

If the velocity of the electron in Bohr s first orbit is 2.19 x 106 ms-1, calculate the de Broglie wavelength associated with it. from Chemistry Structure of Atom Class 11 Haryana
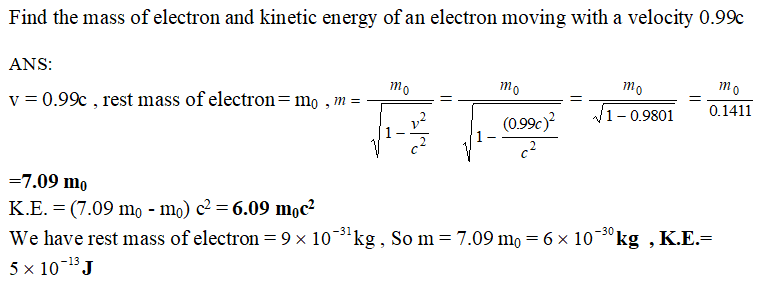
Find the mass of electron and kinetic energy of an electron moving with a velocity 0.99 c : MCQ Answers Guru.PhysicsKerala.in

The ratio of the velocity of light and the velocity of electron in the first orbit of a hydrogen - YouTube
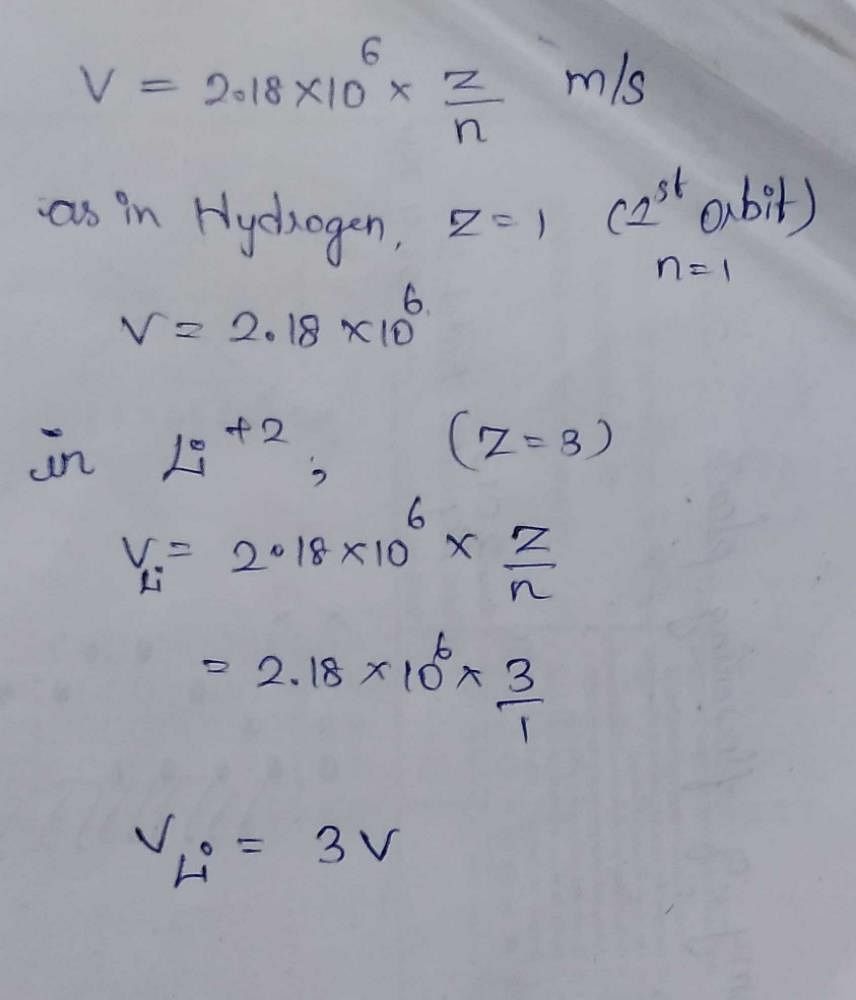
The velocity of an electron in the second orbit of H atom is v the velocity of electron in the first orbit of Li^2 is 1)6v 2)8v 3)v 4)4v? | EduRev NEET

Calculate the velocity of an electron placed in third orbit of H atom .Also calculate of revolatio - YouTube